Project Description
The metal chelating affinity chromatography media supplied by EPRUI Biotech is made up of highly crosslinked monodisperse polyacrylate microspheres.
It is widely used in the purification of His-tagged recombinant proteins. The immobilized metal affinity chromatography resin provides exceptional binding capacity and performance for recombinant tagged protein purification.
The first step of preparing metal chelating affinity chromatography media is forming a hydrophilic layer on the microspheres. Then, nickel ion (Ni2+) functional groups are chelated onto the hydrophilic layer.
EPRUI’s affinity chromatography media consisting of chelating ligands charged with nickel ions are specifically designed to capture and purify histidine tagged proteins.

Applications
Column: 1mL, EPRUI 50Ni-IDA
Sample: His-Tagged protein, Mw=30KD
Binding buffer: 20 mM Tris-HCl, 300mM NaCl, 5 mM Imidazole, pH8.0
Eluent buffer: 20 mM Tris-HCl, 300mM NaCl, 500 mM Imidazole, pH8.0
Flow rate: 1 ml/min
Detection: AKTA Purifier (UV@280nm)
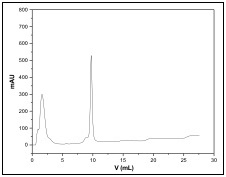
Ni2+ chelating affinity chromatography media is widely used in the purification of His-tagged recombinant proteins as well as peptides, proteins, nucleotides, and phosphorylated proteins which can be adsorbed by metal ions.
Ni-NTA Agarose V.S. Ni-NTA Polyacrylate
| Items | Ni-NTA Agarose | Ni-NTA Polyacrylate |
|---|---|---|
| Matrix | Agarose | Highly crosslinked PMMA |
| Ligand | NTA-Ni2+/IDA- Ni2+ | NTA-Ni2+/IDA- Ni2+ |
| Ligand Density | 20-40umol/ml | 200umol/ml |
| Loading Capacity | 20-30mg/ml | 50mg/ml |
| Particle Size Distribution | 45-165um | 50um |
| Highest flow speed | 500cm/h | 2000cm/h |
| PH range | 3-10 | 3-12 |
| Storage Temperature | 4-8℃ | 4-8℃ |
| Back pressure resistance | 0.1Mpa | 10Mpa |
Features
- High flow rate
- Highly uniform particle size
- Stable chemical property
- Good mechanical stability
- Large specific surface area:600m2/g
- High loading capacity which is two times higher than traditional polysaccharide matrix
- IDA and NTA as affinity media
- Compatible with a very wide range of reducing agents
Product List
| Item | Particle Size | Functional Groups | Ligand | Pore Size |
|---|---|---|---|---|
| EPRUI-Ni50M | 50um | Ni2+ | IDA/NTA | 800A |
